Research
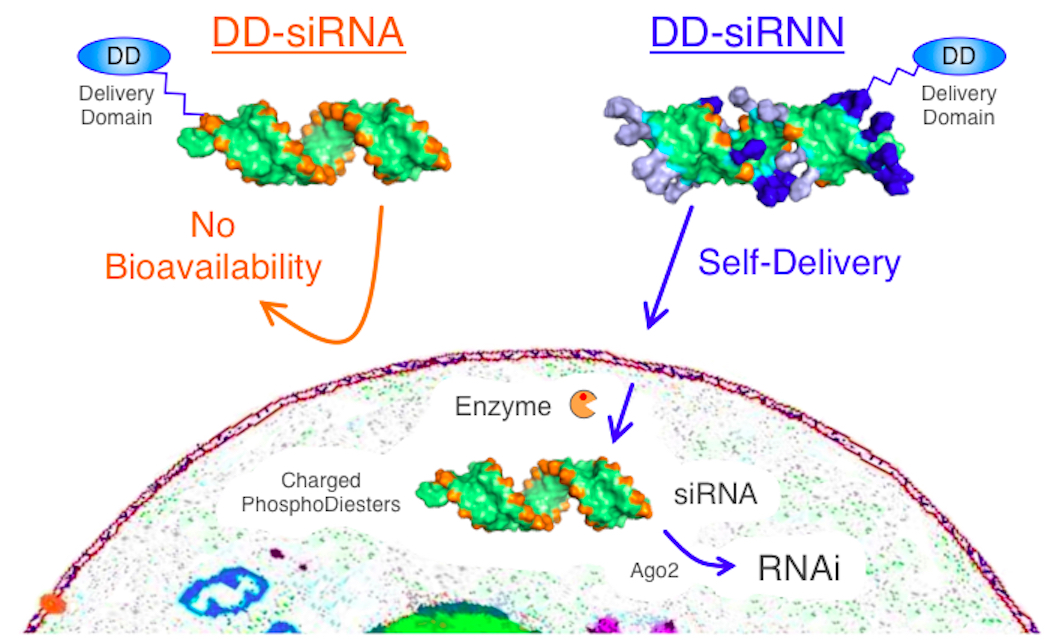
siRNNs prodrug
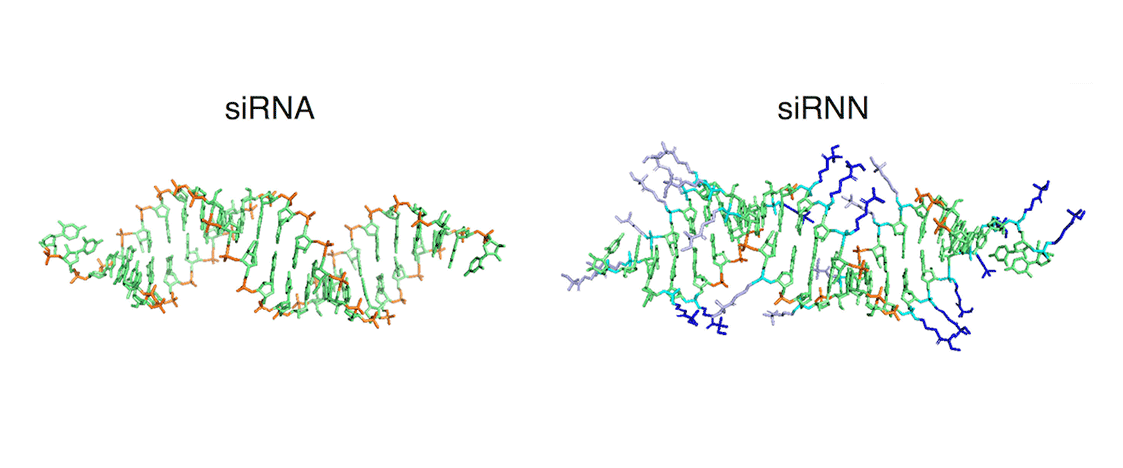
short interfering ribonucleic neutrals (siRNNs) whose phosphate backbone contain neutral phosphotriester groups, allowing for delivery into cells. Once inside cells, siRNNs are converted by cytoplasmic thioesterases into native, charged phosphodiester-backbone siRNAs, that induce robust RNAi responses. siRNNs have favorable drug-like properties, including serum stability and absence of innate immune responses. Unlike highly charged siRNAs, siRNNs also avidly bind serum albumin to positively influence pharmacokinetic properties. Systemic delivery of siRNNs conjugated to a targeting domain induces extended dose-dependent in vivo RNAi responses in mice.
Cell Cycle
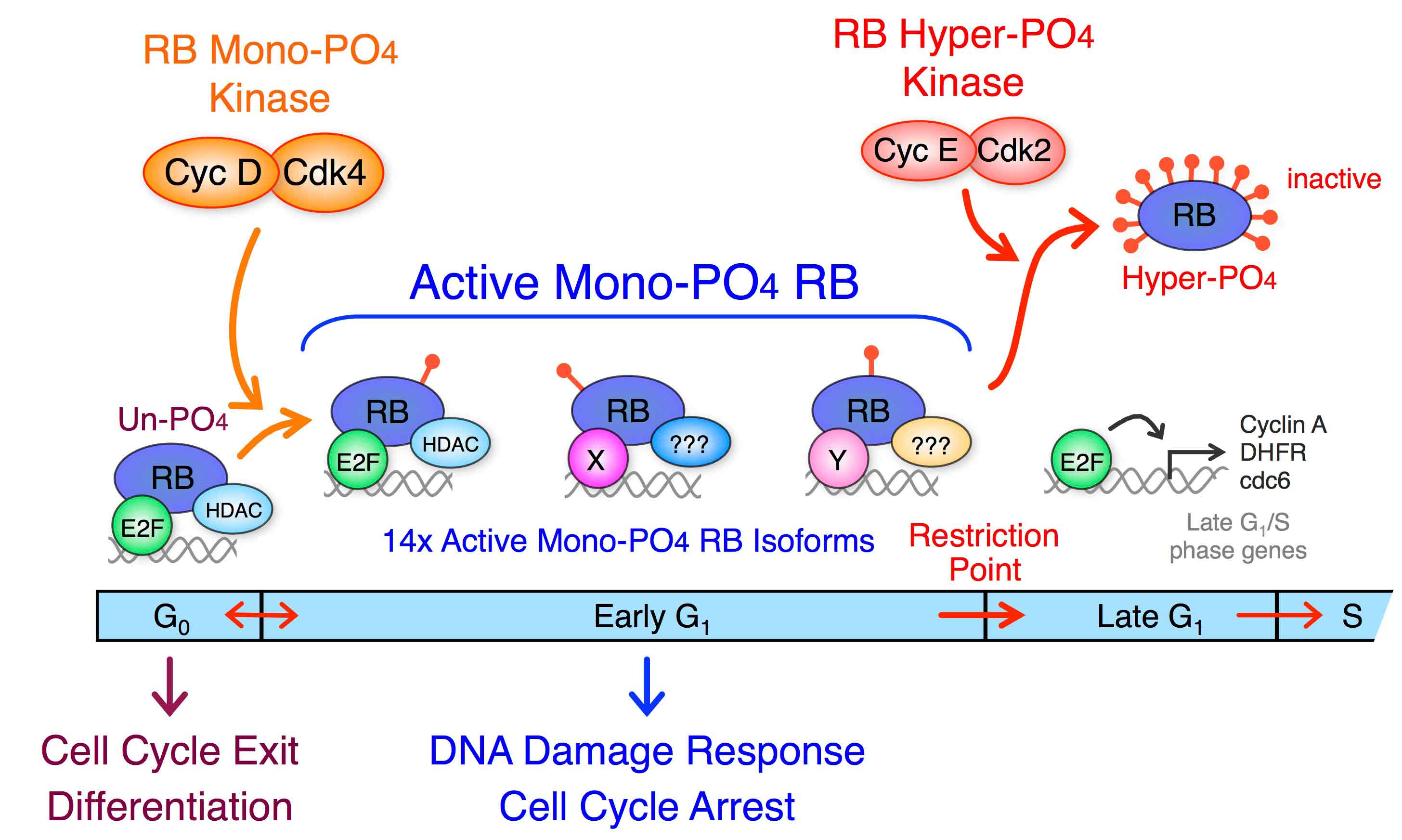
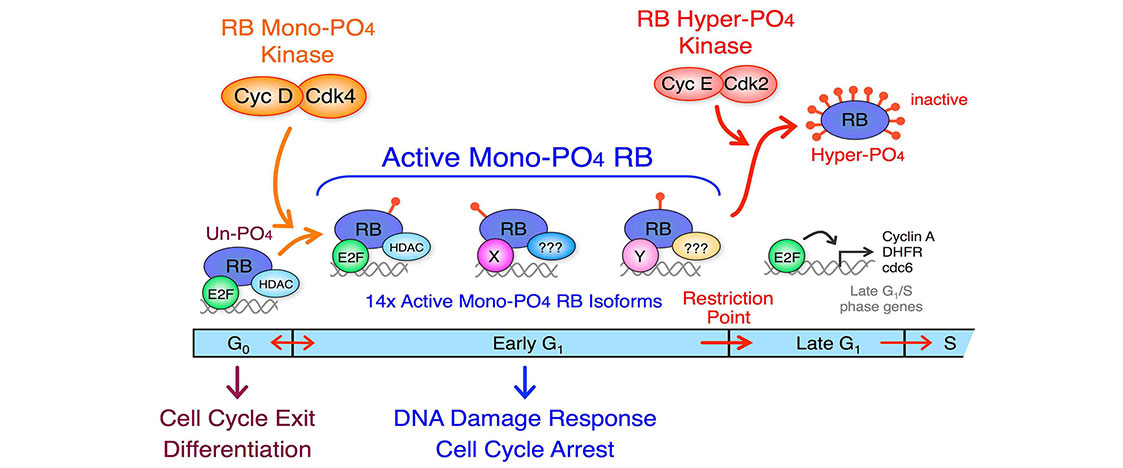
Our lab is interested in the regulation of G1 cell cycle progression in cancer and determining the consequences of these genetic and epigenetic alterations and the mechanisms that activate the cell cycle machinery. We have shown that cyclin D:Cdk4 activates the RB tumor suppressor in early G1 phase by generating 14 individual mono-phosphorylated RB isoforms that each preferentially bind specific and overlapping targets. Activation of cyclin E:Cdk2 at the early to late G1 Restriction Point transition performs the initial RB inactivation by hyper-phosphorylation (14x phosphates per RB). Our goal is to understand the regulatory interplay of activating and inactivating cyclin:Cdk complexes on RB and how these are deregulated in cancer.